
Undergo rearrangement to an aldose and will therefore behave as a reducing sugar. At the very high pH of Benedict's reagent, an alpha-hydroxy ketose, like fructose, can Small amounts of Cu(1) generated by the reaction may produce a green or The Cu(1) product of the reaction forms aĭark red precipitate. Turn, the Cu(II), the oxidizing agent, is reduced to Cu(1). In the Benedict's reaction, the reducing sugar is oxidized by Cu(II). Remember that in an oxidation-reduction reaction, one substance becomes reduced and another One that is bonded to both the ring oxygenīenedict's reagent (CuSO4/sodium carbonate/sodium citrate) is used to detect reducing sugars. Monosaccharides such as glucose and disaccharides such as lactose ('milk sugar') and sucroseīecause they contain a chemically reactiveĬarbon atom (marked with an arrow) is the Polymers (polysaccharides) such as cellulose, starch, and glycogen. Identify solutions of glucose, fructose, lactose, and sucrose.ĭetermine what sugars are present in honey.ĭistinguish pure corn syrup from high-fructose corn syrup.Ĭarbohydrates (also called sugars or saccharides) have the general formula (CH20), and can oftenīe recognized by their large number of hydroxyl (-OH) groups. Samples will give weak positive results that can be useful. Perform will give mostly qualitative (yes/no) rather than quantitative (numerical) results, but some In this lab exercise, you will use three different chemical reactions to detect and attempt to identifyĭifferent types of carbohydrates in pure solutions and in mixtures. 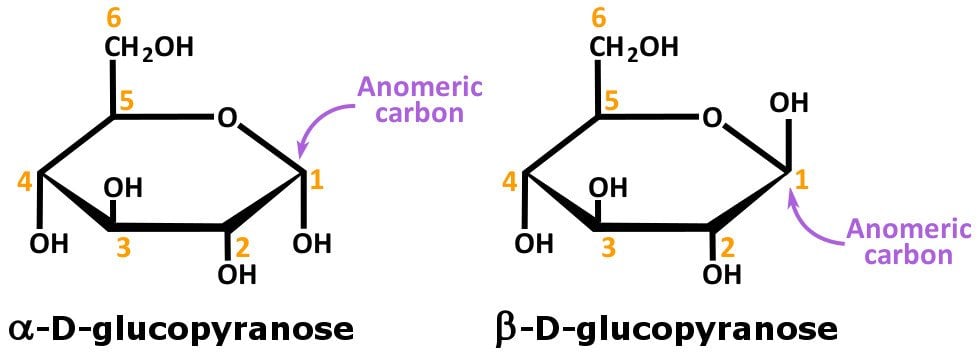
Relate chemical reactivity to structural features of different carbohydrates.ĭetermine the composition of carbohydrate mixtures. Perform qualitative assays for carbohydrates.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
